
A stem-cell based approach against photoaging
We have all heard the term “photoaging” and can agree on its definition, succinctly described by the Canadian Dermatology Association: “Photoaging is premature aging of the skin caused by repeated exposure to ultraviolet radiation (UV), primarily from the sun but also from artificial UV sources. Photoaging differs from chronologic aging: the damaging effects of UV rays – from the sun or artificial tanning sources – alter normal skin structures.”(1) As a result, this UV light causes DNA changes at a cellular level, and because photodamage happens in the deepest layers of the skin – the dermis – it can take years before the damage surfaces and becomes visible(2). (As we all know, UVA radiation is highly damaging, penetrating much deeper than UVB radiation; while UVB causes sunburn, UVA results in premature skin aging and wrinkle formation – and both, of course, can cause skin cancers(3).) Historically speaking, our industry has long focused on protection against UV radiation via the use of sunscreens (which remain on the surface of the skin), but later scientific advances have produced ingredients and formulations intended to penetrate the skin and act to protect cells in the dermal and epidermal layers. And the most recent efforts have been concentrated on the protection of keratinocyte stem cells in the epidermis against UVA-induced DNA lesions, along with boosting the cells’ endogenous DNA-repair system.
It must be mentioned that the concept of “stem cells” commonly brings to mind embryonic stem cells, which can form new cells of any kind, but here we are referring to epidermal stem cells, which can only differentiate into keratinocytes. And as we know from basic skin biology, keratinocytes – if healthy – can potentially renew indefinitely, providing a continuous supply of new cells to the epidermis as part of the essential renewal process of the skin.
But back to the topic of UVA protection: It has been shown that epidermal stem cells have a greater capacity to fight against UVA aggression than their “daughter”, or differentiated, cells, due to more efficient repair mechanisms(4). By the same token, epidermal stem cells are highly sensitive to UVA-induced damage and lose their “stemness” potential when exposed to excessive UVA radiation, which is why it is of utmost importance to protect these stem cells so that they remain robust and productive. It has recently been discovered that an extract from the fruit sechium edule can provide significant biological protection to these stem cells against UVA-induced epidermal damage(5).
The protective effect of sechium edule fruit extract on the epidermis can be observed through its protection against UVA-induced DNA damage, boosting of DNA repair capacities, and maintenance of the stemness potential of keratinocytes.
It was found that exposure to UVA induces the formation of oxidative DNA lesions, specifically 8-oxoG lesions, in keratinocyte cells and causes extensive damage to DNA in the form of fragmentation. A study conducted with the comet assay showcases the severity of UVA-induced damage on DNA as visualized in the samples exposed to UVA with the fragmented DNA forming a tail behind intact DNA. Treatment with sechium edule fruit extract helps to protect keratinocytes by significantly reducing the number of oxidative lesions thereby helping to keep the DNA intact (see Figure 1).
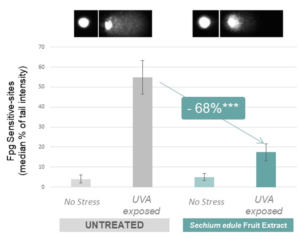
Figure 1. Protection against UVA-induced DNA damage (***p<0.001) (6)
As mentioned earlier, the human body is equipped with its own endogenous repair mechanisms that can repair DNA, but these mechanisms can often be affected by UV and become less effective. The next study looked at the enzymes involved in the repair of 8-oxoG lesions, OGG1 & MYH. The treatment of keratinocytes with the extract brought about a significant increase in the mRNA expression of both OOG1 and MYH suggesting an increase in the DNA repair capacities (see Figure 2).
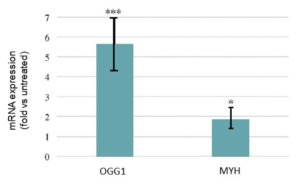
Figure 2. Increase in enzymes associated with DNA repair capacities of 8oxo-G lesions (***: p<0.001; *: p<0.05) (6)
The most important aspect of protection against photoaging is the preservation of the stemness potential of keratinocytes to ensure proper epidermal homeostasis. The last study observed the ability of keratinocyte stem cells to multiply and form new keratinocytes. In the graph below, holoclones refer to cells capable of extensive proliferation and meroclones are cells with limited proliferation capacity. You can think of holoclones as keratinocyte stem cells and meroclones as daughter cells.
When exposed to UVA, the holoclones change into meroclones and the number of colonies drastically decreases, showcasing that the stemness potential has been negatively impacted by UV exposure and the self-renewal capacity lost. In comparison, holoclones treated with the extract of sechium edule fruit and exposed to UVA present a significant increase in number of colonies and the stemness potential of keratinocytes preserved (see Figure 3).
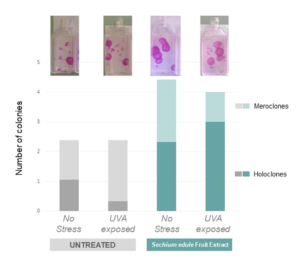
Figure 3. The preservation of stemness potential in keratinocytes (6)
In conclusion, when the “stemness” potential of the keratinocytes is preserved it results in proper skin homeostasis and can lead to improved skin appearance (smoother skin, reduced wrinkles, more even skin tone, etc.). In addition to the use of broad-spectrum sunscreens (always recommended!), an active such as the one described above derived from the fruit of sechium edule is another tool in the arsenal of the formulating chemist in the ongoing fight against the effects of photoaging and UV-damage.
References:
- dermatology.ca
- yalemedicine.org
- uihc.org
- Metral, Elodie, et al., “Keratinocyte stem cells are more resistant to UVA radiation than their direct progeny.” PLOS ONE, 2018 Sept, vol. 13, n° 9, https://doi.org/10.1371/journal.pone.0203863
- Metral, Elodie, et al., “Long-term Genoprotection Effect of Sechium edule Fruit Extract Against UVA Irradiation in Keratinocytes.”, Photochemistry and Photobiology, 2018 Mar; 94(2): 343-350, https://doi.org/10.1111/php.12854
- Metral, Elodie, et al. “Long-Term Genoprotection Effect of Sechium Edule Fruit Extract against UVA Irradiation in Keratinocytes.” Photochemistry and Photobiology, 2018 Mar; 94(2): 343-350, https://doi.org/10.1111/php.12854
![]()
Authors:

Ben Blinder is the Executive Director, Business Operations at Gattefossé USA, with P/L responsibility for the personal care and pharmaceutical business units in the US and Mexico. He is also a founding member of the Advisory Committee on Diversity & Inclusion for Gattefossé in North America. Ben holds a BS in chemical engineering from Lehigh University.

Christina Philips is the Technical Marketing Leader at Gattefossé USA, responsible for providing information on trends and customer insights and technical marketing support for the sales team. She works closely with formulation chemists at the North American Technical Center to provide customers with informative lab sessions and conceptualize sensorial prototypes that highlight the company’s ingredients. She also volunteers as the Director of Empowerment for FOREFRONT Charity. Christina holds a BS in cellular and molecular biology from the University of Connecticut.
Silicone Alternative Solutions for Hair Care
Introduction
Silicones have been widely used in the cosmetic industry for decades. They are exceptionally versatile and impart multifaceted benefits across a wide range of beauty and personal care products. Not all silicones have been created equal, however, and some of these materials are now limited by regulatory restrictions on their use. Due to an increasing focus on their toxicological and environmental impact, consumers are now gravitating towards natural, safe, and sustainable alternatives. This article provides an overview on how silicones have shaped the hair care industry and the continuous research necessary to find innovative and environmentally friendly alternatives to silicones.
Influence of Silicones in the Hair Care Industry
Silicones have been important ingredients in hair care products since the 1950’s. Silicones or silicone derivatives are widely used in shampoos, conditioners, colorants, or styling products where they act as either emollients, humectants, film formers, antifoaming, anti-static, or binding agents [1]. These materials range from basic cyclic or linear polydimethylsiloxane (PDMS) to polyether-and amino-based fluids and silicone resins [2]. All silicones have a natural origin (silica), but synthetic processes are used to create the plethora of silicone ingredients used in cosmetics [3].
Linear PDMS, also known as dimethicone, is available in a range of molecular weights and viscosities and is most used in hair care applications. They provide excellent conditioning and performance which increases with higher viscosity. Use of dimethicone reduces combing forces, provides great sensory benefits like gliding, and adds suppleness to hair.
Phenyl trimethicone is also based on linear PDMS with the addition of phenyl groups [4]. This combination results in a higher refractive index that effectively coats the hair enhancing its shine and leaving hair soft. PDMS polymers are also highly water resistant which makes them effective agents in reducing tackiness of the formulations.
Cyclic polydimethylsiloxane (cyclic PDMS) or cyclomethicones fluids are characterized by ring structures typically containing three to six groups per ring [2]. These fluids decrease combing forces by reducing friction and surface energy [4]. Due to their volatility and fast spreading properties, they provide transient gloss to hair, leaving hair weightless and without any build-up. Cyclomethicones are more compatible with a wider range of ingredients versus linear PDMS.
Silicone gum/fluid blends provide a high level of substantive conditioning and frizz control while imparting a soft and lubricious feel [4]. There are silicones that are modified, like amodimethicones (amine-functionalized silicones) or alkylmethicones (replacing methyl groups on PDMs with alkyl chains) which are widely used in hair care applications as well. Amodimethicones impart specific benefits like color protection, heat protection, repair, reduced flyways, and deep conditioning.
The above-mentioned silicones are non-water soluble, whereas silicone polyethers are a family of water and/or alcohol soluble materials commonly used in shampoo formulations. They provide light to medium conditioning. In addition to acting as emulsifiers or co-emulsifiers, they can be used as resin modifiers to aid curl retention [2].
Moving Away from Silicones
While silicones have been highly effective hair care ingredients providing both functional and enhanced sensorial benefits, there is a movement away from their use due to a variety of reasons. There are long-term effects of silicone such as causing build-up, greasiness, and scalp accumulation [5]. Furthermore, concerns have been raised about their toxicity and effects on the environment.
The Registration, Evaluation, Authorization and Restriction of Chemicals (REACH) assessed the potential environmental effect of cyclic silicones: cyclotetrasiloxane (D4) and cyclopentasiloxane (D5). Based on the evaluation, D4 meets the criteria for identification as a persistent, bio accumulative and toxic (PBT), very persistent, very bioaccumulative (vPvB) substance and D5 meets the criteria for a vPvB substance [6]. After January 31st, 2020, the concentration of D4/D5 in rinse-off cosmetic products placed on the market should be less than 0.1% by weight of either substance. This has now been amended to include cyclohexasiloxane (D6) and is expected to be further restricted to include leave-on products [7].
Linear silicones are also not completely in the clear. They are suspected to be an environmental toxin and to be bioaccumulative. Dimethicone, Dimethicone Copolymer, Polysilicone-15 and other silicones are commonly considered to be microplastics [8,9]. In addition to being non-biodegradable, silicone oils also have an impact on the environment due to their industrial production process which has a high carbon footprint [10].
High performance, Natural and Sustainable Solutions
According to Mintel’s GNPD, between 2016 and 2021 the incidence of “silicone-free” claims for hair care products increased by over 200%. With regulations to control the usage of silicones in hair care products and a growing emphasis on naturality and sustainability, companies are looking for ingredients that serve as silicone alternatives. There is a huge focus on developing efficient and innovative ingredients that offer similar or better functional performance and with a better sensorial profile than silicones. A few ingredient solutions currently offered are highlighted below:
C13-C15 Alkane (plant-derived) is a sustainable natural silicone replacement developed via the fermentation of renewable sugar and grown sustainably without irrigation. This ingredient meets the performance of dimethicone in frizz reduction and color protection. It also matches the performance of amodimethicone in terms of wet/dry combability and provides an excellent sensorial profile. Ethyl Macadamiate is another silicone alternative of macadamia esters. It is biodegradable and provides the same silky, smooth after feel as cyclopentasiloxane [8]. A highly viscous, hydrogenated polyfarnesene presents interesting properties to replace Dimethiconol-based blends [11].
A prime function of silicones is to act as emollients. A vegetable emollient that is readily biodegradable and reduces significant carbon footprint is a perfect ally to protect hair from repeated mechanical stress and perform superior to cyclopentasiloxane [10]. An example is Hydrogenated Olive Oil which is an unsaponifiable squalene from olive oil and hydrogenated castor oil. It is yet another emollient offering to replace silicone and mineral oils that has great application in anti-frizz haircare products [12].
Reduced Silicone Solutions
It is not necessary to exclude all silicone products; cyclic free or synthetic silicones that meet REACH requirements can still be used as alternative solutions. Using low viscosity dimethicone and a mixture of C13-C14 isoparaffin can be considered as a replacement for cyclomethicones delivering a similar sensory profile [7]. Use of terminal hydroxy amino-modified silicone (THA) chemistry provides long-lasting conditioning and protects hair against breakage [13]. As for synthetic offerings, combining quaternary conditioning properties of cetrimonium chloride with a carboxylated silicone provides thermal protection and enhances the manageability of hair, while a complex of cetrimonium chloride with a water-soluble silicone provides great hair care benefits in different formats [7]. A combination of natural oils and a synthetic polymer can enhance and extend the benefits of natural oils to smooth and restore damaged hair and protect and reduce hair damage from different grooming regimens [12].
The industry also offers some unique solutions like quat-free polymeric conditioning additives that can provide multi-functional benefits to hair which are ideal for amodimethicone free formulations. Lastly, to reduce the carbon footprint, manufacturers are cutting down the high use of energy to produce dimethicones of various viscosities by using methanol obtained from biomass instead of fossil fuels [7].
Using Digital Tools to Source Ingredients
In the quest for clean and sustainable ingredients, Artificial Intelligence (AI) is playing a critical role in research and development. Machine learning is a powerful tool that can collect large amounts of data and provide detailed information about ingredient sourcing [11]. AI will be further integrated to explore unmet needs and help screen and identify innovative ingredients for various applications. Companies are also developing apps, using QR codes to trace ingredients, and promote ingredient transparency by providing origin and sustainable properties [11].
Conclusion
Over the last decade, there has been a strong shift in consumer product preferences with emphasis on personal wellbeing and the environment. The cosmetic industry has made great progress in offering many eco-friendly, clean, and sustainable solutions not only to replace silicones but also other ingredients that are currently being challenged. Companies are tasked to continue their efforts in developing eco-friendly and sustainable products that are highly effective in functional and sensorial performance to meet consumer needs.
References
- Kostic A, “Silicones in cosmetics and their impact on the environment”, Cos ACTIVE J. 2021;1:34–39
- Katie Schaefer, “Silicones in Hair Care: Making Innovative Solutions Possible”, Cosmetics & Toiletries (November,2008)
- Megan McIntyre, “Do Silicones Deserve Their Bad Rap? “, Beauty Independent, June 2019
- Bethany Johnson, Kevin Murphy and Feifei Lin, “How Silicones Shape the Hair Care Industry: A Review”, Cosmetics & Toiletries (June,2015)
- Solvay.com, “How Sulfate and silicone Alternatives Improve the Hair care Industry and Benefit Consumers”
- Mojgan Moddaresi, “Regulation Update: Cyclosiloxanes in the EU”, UL Prospector (February ,2018)
- Smooth closer: The latest in silicones and silicone alternatives”, Cosmetics Business (November,2021)
- SuperZero.com, “What are silicones and why are silicones used in the beauty industry? (April,2021)
- Plastic-TheHiddenBeautyIngredients.pdf (beatthemicrobead.org)
- “An alternate to silicone for hair care”, Personal Care Magazine (March,2019)
- “New generation of emollient showing promising results as a sustainable alternative to viscous silicones in hair care formulations: Seppic”, Cosmetics & Toiletries, Vol.137, No.6 (June 2022)
- John Woodruff, “Silicones and Alternatives 2018”, published by SPC2018
- Nisaraporn Suthiwangcharoen, Bethany Prime, Beth Johnson, and Dawn Carsten, “Simple and Sensorial Amino-modified silicone protects and revives hair”, Cosmetics & Toiletries, Vol.136, No.2 (February 2021)
![]()
 Mythili Nori has worked in the Personal Care industry for over a decade. Her expertise is in Product Claim Substantiation and Data Science. In her current role at BASF, she is responsible for Physical Claim Substantiation & Sensory testing for Hair & Skin Care. Prior to joining BASF, she spent 5 years at TRI/Princeton as a Senior Research Associate, supporting claim substantiation and fundamental research activities for textile and hair surfaces. She earned a Bachelor of Technology in Chemical Engineering from India and received Master of Science in Chemical Engineering at North Carolina Agriculture & Technical State University focusing on purification of drinking water.
Mythili Nori has worked in the Personal Care industry for over a decade. Her expertise is in Product Claim Substantiation and Data Science. In her current role at BASF, she is responsible for Physical Claim Substantiation & Sensory testing for Hair & Skin Care. Prior to joining BASF, she spent 5 years at TRI/Princeton as a Senior Research Associate, supporting claim substantiation and fundamental research activities for textile and hair surfaces. She earned a Bachelor of Technology in Chemical Engineering from India and received Master of Science in Chemical Engineering at North Carolina Agriculture & Technical State University focusing on purification of drinking water.
PPARs reemerging as a skin wellness target in cosmetics
PPARs reemerging as a skin wellness target in cosmetics
Written by Marc Cornell, Mar-Key Consulting LLC
Cosmetics are continuing their marvelous evolution with new scientific findings and breakthrough innovations. Sometimes we need to look back to find the genesis of a current marketing or positioning trend. Take a step back with me to the late 20th century, where a young researcher (MC) was being educated in the biology of wound healing. During this time, I became aware of Peroxisome proliferator-activated receptors (PPARs). PPAR-alpha expression plays a role in the inflammatory stage of wound healing. Later I learned wound healing shares many biologic pathways with anti-age biology. Peroxisome proliferator-activated receptors is a mouthful, right? Let’s dial it back bit and keep the PPAR focus on our body’s largest organ, the skin.
PPAR’s are ligand inducible transcription factors belonging to the nuclear hormone receptor superfamily. The key isoforms of PPARs are notated as (alpha), (beta/delta) and (gamma). These forms can up regulate or down regulate many cellular and metabolic processes (cellular differentiation, proliferation, lipid homeostasis and energy metabolism). These functions are critical to our body and its skin’s immune system, epidermal barrier function and development of pro-inflammatory signals. Peroxisome proliferator-activated receptors (PPARs) are also involved in regulating glucose, lipid homeostasis and modulate mitochondrial function. (1) It is obvious that regulation of PPAR signaling to various biological systems is an opportunity for medicine and cosmetic science
In some publications, regulation of PPAR signaling have been linked to disease pathogenesis. These ailments have complex causes involving genetic, environmental, and nutritional factors, PPARS are just a part of the pathway. (2) In cosmetic science we are careful to draw the line between drug and cosmetic claims. With that said, there are numerous scientific studies elucidating shared connections between drug and cosmetic active biologic modes of action.
Now we take a partial deep dive on the FAQ’s for PPARS. From there we will highlight how targeting PPAR’s has been around for some time in cosmetics. More recently these chemistries are trending in a big way as clinical branded cosmetics remerge as part of the wellness positioning.
How can cosmetic science leverage PPARS to facilitate skin wellness? We start our PPAR discussion by looking at the skin barrier function. This area of skin biology is also part of a recent trend towards positioning and the minimization of environmental stressors on the exposome. The exposome is defined as the measure of all the exposures of an individual in a lifetime and how those exposures relate to health. PPAR activation has been shown to have an important role in skin barrier function by regulating differentiation and lipid synthesis in keratinocytes. (3) This fact fits nicely into homeostasis wellness positioning.
PPAR activation takes place through heterodimerization. Simply put, you need 2 ligands to come together on the DNA to initiate gene regulation. PPAR activators are already being used in areas of cosmetic science application. Botanical extracts have been shown to activate PPAR in the stimulation of collagen in skin (4). There is also evidence that PPARs may be used as an alternative to retinoids in skin care. (5) PPARs have an important effect in keratinocyte homeostasis, suggesting a role in diseases such as psoriasis. (6) PPAR acts directly to negatively regulate gene expression of proinflammatory genes in a ligand-dependent manner by antagonizing the activities of transcription factors such as members of the NF-kB and AP-1 families. (7). One very popular cosmetic application is the use of omega 3 fatty acids. These are popular natural ligands for PPARα receptors and are key to preventing/reducing inflammation.
One critical factor in attempting to utilize pathways linked to PPARs is in the understanding of the up or down regulation of these biochemicals. Typical of any biologic system there is potential for biofeedback, so understanding the pathways and assay methodologies are important. Good news is gene expression and cell culture models now allow the high throughput analysis of materials which may take part in PPAR regulation. This fact and the numerous peer review publications give the cosmetic chemist a boost in the study of PPARs.
In closing I provide just a few key words for continued education on PPARs.
Links between PPAR and:
a) Endocannabinoid receptor system
b) Hyaluronic acid
c) Wound healing
d) Retinoid receptor system
e) UV modulated inflammation
f) Inflamaging
g) Mitochondrial function
h) and MORE!
- Ting-Wei Lee, Kuan-Jen Bai, Ting-I Lee, Tze-Fan Chao, Yu-Hsun Kao & Yi-Jen Chen : PPARs modulate cardiac metabolism and mitochondrial function in diabetes Journal of Biomedical Science volume 24, Article number: 5 (2017)
- Kersten S, Desvergne B, Wahli W. : Roles of PPARs in health and disease. Nature. 2000; 405: 421–4. NATURE
- Su-Hyoun Chon , Ruth Tannahill, Xiang Yao, Michael D Southall, Apostolos Pappas. Keratinocyte differentiation and upregulation of ceramide synthesis induced by an oat lipid extract via the activation of PPAR pathways , Exp Dermatol. 2015 Apr;24 (4):290-5
- George P. Majewski1 | Smrita Singh2 | Krzysztof Bojanowski :Olive leaf-derived PPAR agonist complex induces collagen IV synthesis in human skin models, Int J Cosmet Sci. 2021;43:662–676
- John Simon Craw, George Majewski: Coding Skin for Care: PPAR Ligands as Retinoid Alternatives and Adjuvants—Cosmetic and Toiletries, Jan 6th, 2022
- Emerson de Andrade Lima1 et al : Peroxisome proliferator-activated receptor agonists (PPARs): a promising prospect in the treatment of psoriasis and psoriatic arthritis* An Bras Dermatol. 2013;88(6):1029-35.
- Yuval Ramot et al, The role of PPAR-mediated signaling in skin biology and pathology: new targets and opportunities for clinical dermatology : Experimental Dermatology, 2015, 24, 245–251
![]()
Written by Marc Cornell, Mar-Key Consulting LLC

Marc Cornell, BS. is a consultant at Mar-key Consulting LLC where he services the consumer product industry with innovative formulation concepts. During his thirty-year career he has worked in an R&D role for large (Merck, L’Oreal, Bristol Meyers Squibb, Union Carbide) and medium sized companies (Neostrata, ChemAid Labs, KV Pharmaceutical). For the last 20 years he has worked primarily on the research and formulation development of “Cosmeceuticals” for various brands (Skinceuticals, Neostrata, Dr. Perricone, Biomedic, Strivectin, and La Roche Posay). In this role he collaborated with researchers in skin biology and clinical testing to design, formulate and test novel cosmetic active delivery vehicles. Marc’s work has been patented and published in peer review journals and trade publications.
Natural Ingredients in Cosmetics
Natural ingredients offer a myriad of possibilities for developing effective cosmetic products. Their popularity has greatly increased over the past two decades in part due to a major shift in public opinion about the environment, human health, and wellbeing. Plant ingredients have been shown to be effective treatments of the skin for a number of conditions including erythema, hyperpigmentation, photoaging, photocarcinogenesis, and photoimmunosuppression. Nowadays, botanical ingredients are found in almost every type of cosmetic product for the skin. In addition to plants, minerals are also natural ingredients. Some of the most common ones found in modern-day cosmetic products consist of iron oxides, zinc oxide, and titanium oxide, which are mostly used in sunscreen formulations.
Historical Perspective of Natural Ingredients in Cosmetics
Natural ingredients have been used in cosmetic products since antiquity. The early Egyptians were renowned for their makeup preparations and other cosmetic ingredients used to cleanse and scent the body. The most common cosmetic potions consisted of eye paints, facial paints, oils, and solid fats (ointments) [1]. As an example, kohl is a paste/powder that was commonly used as eye shadow and is reported to have been made with galena ore, which contains lead sulfide. A paste made from malachite, a green ore of copper, was also used to color the eyes of Egyptians. Some of these ingredients probably caused adverse reactions, or could have led to serious disease after prolonged use.
Hair and nail dyeing in ancient Egypt was achieved using henna, which was extracted from the plant Lawsonia inermis, also known as the Egyptian privet. Henna was also popular in ancient India and China as a hair dyeing agent. In India, henna was also used to paint designs on the hands and feet in the art known as mehndi [2]. The early Egyptians also used fats and oils to apply to skin and hair, protecting them from the powerful sun rays and arid climate. The Egyptians were also very astute on the use of fragrances. They used many different types of herbs and oils, such as aloe, chamomile, lavender, myrrh, olive oil, peppermint, sesame oil, and thyme [3].
Turmeric, a traditional Indian spice from the root of Curcuma longa, was commonly used in Ayurvedic medicine as a therapeutic agent. It contains curcumin, which has anti-inflammatory properties. In recent years, turmeric has become an extremely popular cosmetic ingredient for skin care preparations. In traditional Chinese culture, skin was treated oils and herbs. Panax ginseng is one of the most popular ingredients in ancient herbal therapy, and is still widely used today. Rice powder was also popular and used to paint the face, serving as a form of makeup that provided a whitish appearance and had the benefit of removing excessive oils. The use of nail polish dates back to ancient China, using egg whites, flowers, and beeswax [4]. Unfortunately, not all members of society were permitted to paint their nails. It was reserved for royals, who painted their nails gold and silver, and other members of the upper echelon of society.
Natural ingredients were also used in cosmetics in other periods of history as well. In biblical times, the Hebrews used oils obtained from various plant and animal sources as emollients to protect the skin from the arid environment and intense solar radiation. In addition, red ochre (an iron oxide) was used for painting the lips, ash and beeswax for painting the nails, and herbal perfumes were applied to the skin and clothing [5]. During the early Roman Empire, Pliny the Elder (Gaius Plinius Secundus), who was a prolific author, naturalist, and philosopher, wrote about the control of perspiration using a mixture of rue, rose oil, and aloe vera [6].
In the western tradition, the use of natural ingredients in cosmetics continued through the Middle Ages and Renaissance all the way to the 19th century, although the overall use of cosmetics fluctuated throughout history most likely due to sociological and economic factors. Curiously, at the dawn of the 20th century, color cosmetics were not very popular in western societies, and even frowned upon for women to wear in public. In the United States and many other western cultures, this attitude began to change significantly as movie stars began wearing makeup products in Hollywood films. During this period, there was a flurry of activity in the development of highly functional synthetic ingredients that enjoyed widespread use in cosmetic products. However, the most recent natural ingredient movement began to take place in the late 1990s and early 2000s as the population became more concerned with health, wellbeing, and global environmental conditions.
Botanical Ingredients
The increasing awareness of the health benefits of phytochemicals has led to a transformation in the cosmetic industry [7]. The recent explosion of the use of herbal ingredients in cosmetic products began with ingredients that offered improvement in the physiological condition of the skin by treatment with formulas containing plant ingredients [8]. This movement evolved to include a greater effort to replace conventional synthetic ingredients that carried other functions in the formula, such as rheology modifiers, emollients, cleansing agents, etc. [9]. Today, there are even some forms of cosmetic packaging that are based on natural or naturally derived ingredients.
There are numerous plant ingredients that are used in cosmetic products for their cosmeceutical properties. Some of the most common ingredients include Aloe vera, Camellia sinensis (tea polyphenols), Capparis spinosa flower buds, Culcitium reflexum H.B.K. leaf, Curcuma longa (curcumin), French maritime pine bark (pycnogenol) Gingko biloba, pomegranate fruit, red orange, Sanguisorba officinalis L. root, Sedum telephium L. leaf, and Silybum marianum (Silymarin). Extracts of natural products contain polyphenols and other phytonutrients that have beneficial effects for the skin. Plants evolved to produce these ingredients to protect themselves from environmental insults, including harmful UV radiation.
Many botanicals have been used for millennia in traditional Chinese medicine and Ayurveda. Nowadays, there is a flurry of activity in the skin care market with similar types of ingredients, due to a growing body of scientific evidence demonstrating their utility as skin therapeutic agents. Among other things, botanical ingredients have shown promise as anti-inflammatories for skin to treat rosacea, preventative agents against melanoma, bioactives for the treatment of skin aging, and protective agents against UV-induced immunosuppression and photocarcinogenesis [10].
Incorporating plant ingredients into cosmetics can also present challenges to the formulator in terms of stability and delivery [11]. For this reason, there have been many efforts focused on developing carrier systems for botanical ingredients [10, 12]. Most of these carriers are emulsions, vesicular systems, or lipid particulate systems. Emulsions for this type of application usually are microemulsions, nanoemulsions, micro-nanoemulsions, multilayer emulsions, or Pickering emulsions. Common vesicular systems consist of liposomes, ethosomes, phytosomes, and transferosomes. The two most popular lipid particulate systems are solid-lipid nanoparticles and nanostructured lipid carriers.
Polysaccharide Ingredients
Polysaccharides from many natural sources are used in cosmetics. They are often added to formulas as rheology modifiers, but may also be used for a variety of other functions, such as providing moisture to the skin or enhancing the styling properties of hair. The most common polysaccharides found in cosmetic products are agar, alginate, carrageenan, derivatives of cellulose (e.g., hydroxyethylcellulose), chitin, chitosan, dextrin, guar gum derivatives, gum arabic, hyaluronic acid, pectins, starch derivatives, and xanthan gum. In addition to the applications already mentioned, polysaccharides are also found in masks and shampoos/body washes (coacervate agent). A number of different polysaccharides may also be included in personal care products for their antibacterial, antiviral, anticoagulant, anticancer, antioxidant, and immunomodulating activity [13]. Overall, they have a long and safe history of use in cosmetic products.
Essential Oils
Essential oils enjoy widespread utility in cosmetic products due to their pleasant odor and biological activity [14-16]. They are highly concentrated liquid mixtures of small molecules (mostly aromatic compounds, terpenes, and terpenoids) extracted from the bark, buds, flowers, fruits, leaves, rhizomes, roots, and seeds of plants [14]. Some of the most common essential oils found in cosmetic products are citronellol, citrus, eucalyptus, geraniol, lavender, limonene, linalool, and tea tree [16]. If formulated at low concentrations, essential oils are relatively safe. However, at higher concentrations their use may result in skin sensitivity reactions and even the development of allergies [15]. In addition to their aromatic characteristics, essential oils have analgesic, antibiotic, and antiviral properties. For this reason, there is a great deal of interest in aroma therapy and its positive health benefits.
Toxicological Considerations
There is some concern about the safety and toxicology of natural ingredients. This mostly stems from the presence of ingredients that are not listed on the labels of cosmetic products. For example, citral, farnesol, limonene, and limanol—fragrance compounds present in many natural ingredient products—can illicit allergic reactions [17]. Furthermore, there could be many molecules in the formula that are only listed as one ingredient. On the other hand, it has been argued that exposure to natural toxic substances in personal care products is probably not the principal route of exposure. Rather, direct exposure to vegetation and agricultural crops is considered the most dominant pathway [18]. Skin sensitization is another concern with the use of botanical ingredients [19]. As an example, the Feverfew plant (Tanacetum parthenium), known for its anti-inflammatory properties, contains parthenolide, which is a potent skin sensitization agent. Therefore, being able to produce parthenolide-free bioactives is a key challenge to provide a non-sensitizing product for skin care [20, 21].
Concluding Remarks
Natural ingredients have a long history in cosmetics products. Overall, there has been a great deal of renewed interest in their inclusion in contemporary personal care formulas. Combined with modern analytical and process technology, today’s cosmetic chemist has the opportunity to participate in the large-scale transformation of the personal care industry.
References
- Lucas, A., Cosmetics, perfumes, and incense in ancient Egypt. J Egypt Arch, 1930. 16(1/2): p. 41-53.
- Nayak, M. and V. Ligade, History of cosmetics in Egypt, India, and China. J Cosmet Sci, 2021. 72: p. 432-441.
- Chaudhri, S. and N. Jain, History of cosmetics. Asian J Pharm, 2009. 3(3): p. 164-167.
- Madnani, N. and K. Khan, Nail cosmetics. Indian J Dermatol Venereol Leprol, 2012. 78: p. 309-317.
- Parish, L. and J. Crissey, Cosmetics: A historical review. Clin Dermatol, 1988. 6(3): p. 1-4.
- Bostock, J. and H. Riley, Rue: eighty-four remedies, in Remedies derived from the garden plants. 1855, Taylor and Francis: London, UK.
- Dini, I. and S. Laneri, The new challenge of green cosmetics: natural food ingredients for cosmetic formulations. Molecules, 2021. 26: p. 3921.
- González-Minero, F. and L. Bravo-Díaz, The use of plants in skin-care products, cosmetics, and fragrances: Past and present. Cosmetics, 2018. 5: p. 50.
- Bom, S., M. Fitas, A. Martins, P. Pinto, H. Ribeiro, and J. Marto, Replacing synthetic ingredients by sustainable natural alternatives: A case study using topical O/W emulsions. Molecules, 2020. 25: p. 4887.
- McMullen, R., Antioxidants and the Skin. 2nd ed. 2019, Boca Raton, FL: CRC Press.
- Hoang, H., J. Moon, and Y. Lee, Natural antioxidants from plant extracts in skincare cosmetics: recent applications, challenges, and perspectives. Cosmetics, 2021. 8: p. 106.
- Yang, S., L. Liu, J. Han, and Y. Tang, Encapsulating plant ingredients for dermocosmetic application: An updated review of delivery systems and characterization techniques. Int J Cosmet Sci, 2020. 42: p. 16-28.
- Ahsan, H., The significance of complex polysaccharides in personal care formulations. J Carbohydr Chem, 2019. 38: p. 213-233.
- Abate, L., A. Bachheti, R. Kumar Bachheti, A. Husen, G. M, and D. Pandey, Potential role of forest-based plants in essential oil production: An approach to cosmetic and personal health care applications, in Non-Timber Forest Products: Food, Healthcare and Industrial Applications, A. Husen, R. Kumar Bachheti, and A. Bachheti, Editors. 2021, Sprinter Nature: Cham, Switzerland. p. 1-18.
- Sarkic, A. and I. Stappen, Essential oils and their single compounds in cosmetics—A critical review. Cosmetics, 2018. 5: p. 11.
- Sharmeen, J., F. Mahomoodally, G. Zengin, and F. Maggi, Essential oils as natural sources of fragrance compounds for cosmetics and cosmeceuticals. Molecules, 2021. 26: p. 666.
- Klaschka, U., Natural personal care products—analysis of ingredient lists and legal situation. Environ Sci Eur, 2016. 28: p. 8.
- Bucheli, T., B. Strobel, and H. Hansen, Personal care products are only one of many exposure routes of natural toxic substances to humans and the environment Cosmetics, 2018. 5: p. 10.
- Puginier, M., A. Roso, H. Groux, C. Gerbeix, and F. Cottrez, Strategy to avoid skin sensitization: application to botanical cosmetic ingredients. Cosmetics, 2022. 9(2): p. 40.
- Koganov, M., Parthenolide free bioactive ingredients from Feverfew (Tanacetum parthenium) and processes for their production and use. U.S. Patent No. 7,537,791. 2009.
- Sur, R., K. Martin, F. Liebel, P. Lyte, S. Shapiro, and M. Southall, Anti-inflammatory activity of parthenolide-depleted Feverfew (Tancetum parthenium). Inflammopharmacology, 2009. 17(1): p. 42-49.
The use of in-vitro modeling to predict clinical outcome
The use of in-vitro modeling to predict clinical outcome: A users guide to technology development
In-vitro models have been a staple of technology development as well as a tool for mapping mechanistic understanding of skin biology. Their complexity has evolved over time to become quite predictive to clinical outcomes. In-vitro modeling is a powerful solution for driving product development, innovation and claim substantiation, and it allows researchers to screen technologies as well as position them against new or novel mechanisms. The NYCSCC is offering a mini-symposium at this year’s Supplier’s Day (May 3rd and 4th) to present various in-vitro models currently available to our industry using a cross section of speakers from both academia, industry suppliers and testing firms. (See the end of the blog for a list of topics being presented.) The following is a good primer for those interested in leveraging in-vitro data for technology development.
In-vitro models are the result of many of the inventions we learned about in science class at an early age. Here is a brief history of how we got here: First was the invention of the microscope by A. Van Leeuwenhoek in 1676, then Schleiden and Schwann’s formulated cell theory in 1839, Roux’s first method of cell culture in 1885, aseptic techniques for cell culture in 1912, establishment of the first mouse fibroblast cell in 1943, the first immortal human cell line known as “HeLa” by George Gey in 1951, the finite life span of human cells defined by Hayflick in 1965, the first therapeutic protein manufactured in cell culture for human clinical trial in 1983, culture human embryonic stems cells isolated by Thomson & Gearhart in 1998 and finally 3D tissue and organ bioprinting techniques introduced in 2010.
At present, it is no longer possible to use animal testing for ingredients and cosmetic products in the Organization for Economic Co-operation and Development (OECD) member states since 2009. This mandate has served as an “evolutionary pressure” on the industry to leverage in-vitro modeling as an alternative to animal use in in our industry, leading to the development and moderate acceptance of in-vitro tests that are used to determine the safety and efficacy of ingredients and topical products.
The advantages of in-vitro testing are:
1) Reducing the use of human subjects in the early stages of R&D projects; this not only has logistical and ethical benefit in terms of time and money but also removes the regulatory and safety aspects when discovery is the main strategic driver.
2) In-vitro modeling can be designed for high throughput screening of compounds/technologies. As a result, modeling this way saves time and money.
3) Reducing clinical variability as conditions are better controlled and repeatable.
4) Little to no testing regulations; however, sound experimental design including appropriate controls, benchmarks, time points and culture conditions are paramount to success.
The most saliant disadvantage of using in-vitro testing models is that results and findings need to be confirmed in/on more complex systems. For example, if you screen for gene effects in monolayer cultures, one will need to confirm protein expression to confirm the gene expression profile. Furthermore, more complex culture systems such as 3D culture models or ex-vivo biopsy modeling or human testing would be the logical next step to evaluate bioavailability, bioconversion, and efficacy.
The latter would require an intermediate step of safety assessment, ethics review (IRB) and cost analysis. Furthermore, in-vitro modeling does require dedicated space and personnel. This is certainly a capital expenditure; however, there are 3rd party laboratories and collaborations with academia available for almost every need if you look hard enough.
The ability to customize a culture system to fit one’s needs is very encouraging. In-vitro models range from simple monolayer systems to complex muti-tissue engineered models. Advances in maintaining cells in culture are continuously improving, allowing for data generation that closely mimics clinical results. The advent of bio chambers, flexible substrates and precisely defined medias have advanced the understanding of cell biology exponentially. The technics for retrieving and cultivating keratinocytes, fibroblast, melanocytes and dendric cells from skin are commonplace in cell culture facilities around the globe resulting on less reliance on transformed cell lines (those that are immortal) which have a poor correlation to clinical outcome in many cases. 3D skin models with natural human features are best used to analyze ingredients and formulations for this reason. Skin irritation and toxicity tend to be represented by less stringent biomimicry, but this too is evolving to more relevant culture systems.
The vast number of possible applications and products in our industry can target different body areas including intact skin, thin skin (under eye), oily skin (face vs abdomen), sun exposed vs sun protected, healthy or impaired skin which come with many specific characteristics in composition, structure, and function as a primary focus.
In-vitro reconstructed human tissue models are recognized as being sensitive and reliable for areas outside of skin in a pre-clinical environment. Using 3D human reconstructed models such as gingival and vaginal systems can model mucosal tolerance, “immature” epidermal models can mimic infant/baby skin, and culture conditions can be designed to impair function (missing ceramide production) or condition (sensitive skin) to find solutions to the impairment such as in barrier function.
Topical applications represent a vast range of formulation options, accounting for multiple product types focusing on health, gender, age conditions, and body targets. The constant quest for innovation, whether through the search for new molecules or the extended use of old ones, pushes our industry to take many factors into account in the early stages of development. Multiple models of different variations can be applied in sequence to the same project scope representing multiple but independent evaluations and approaches simultaneously.
Manufacturers are also looking to in-vitro modeling to satisfy environmental impact effects of their ingredients and products. Reef safe, biodegradability and species toxicology are becoming commonplace in raw material questionnaire surveys and in environmentally responsible platforms within many companies.
The list of in-vitro models is endless; however, there are a handful of go-to models that make up many R&D arsenals. Reconstructed human epidermis (RHE) is a very common model that uses keratinocytes harvested from surgically excised tissue. When these cells are cultured at the air/media interface of a culture well, they naturally form a representative viably functioning epidermis. Full thickness skin models (FTSK) are composed of both a dermal compartment containing human skin fibroblasts embedded in a collagen matrix and human keratinocytes seeded on top to form the epidermis. Ex-vivo skin explants use full thickness whole skin biopsies in culture, which allows the effect of individual ingredients and formulations, transdermal delivery, topical penetration, and percutaneous absorption to be tested in an environment more closely mimicking normal skin. Even though this model system is the closest to intact skin, it has its limitations. Recent in-vitro modeling of neurodermatology has been achieved by generating a co-culture system of sensory neurons and skin cells, which can simulate in vivo human skin and provide innovative solutions to neuro sensory conditions such as itch, neuro inflammation and pain. Pigmented epidermis model composed of normal human keratinocytes cultivated in the presence of melanocytes of phototypes in the basal layer resulting in a gradient of melanin production when exposed to varying degrees of UV, IR and HEV light. Atopy skin models recreate conditions of this disease state via a co-culture of keratinocytes, fibroblasts, dendritic cells, and T-cells which can be applied to screen therapeutics and to gain a better understanding of the biological mechanisms involved.
The future state will focus on using pluripotent stem cells in a variety of culture conditions and matrixes to reconstitute tissues of interest, thus, more accurately modeling solutions for aged, damaged, or dysfunctional tissue. The idea of in-situ (to examine the phenomenon exactly in place where it occurs) bioprinting is already being considered for as a research tool for therapy and the results obtained in that area seem to be promising. It is possible that soon, use of skin bioprinters will be a useful tool in surgical reconstruction and a preferred form of therapy in wound and burns treatment. Alternatively, the idea of bioprinting on a cellular level can further the customization of in-vitro modeling. The possibilities are endless.
![]()
Mini-Symposium: “In-Vitro Modeling to Predict Clinical Outcome”
Michael Anthonavage, Moderator
Presentation topics
- 3D Tissue Model applications: Genetically engineered 3D skin models for the development of cosmetic products and pharmaceutical drugs Research Focus: Human 3D tissue models
- Innate Immunity modeling: Testing the Skin’s Innate Immune Response via In-Vitro and Non-Invasive Clinical Testing Methods
- Sunscreen modeling: Bioequivalence Efficacy Test for Sunscreens: Alternative SPF Test Methods Validation
- Use of in-silico/vitro modeling for barrier and moisture: Skin Barrier and Hydration: Solutions from in silico & in vitro testing to clinical bioanalysis and imaging
- Sensorial modeling in-vitro: The interest of sensorial evaluation in cosmetics: integration of the sensory neuron compartment on unique in-vitro models.
- Bioavailability: In-vitro Studies of Skin Deposition & Permeation and Their Practicality
- Delivery modeling: Advanced Delivery Modeling Techniques – A Runway to Success
- Total exposure modeling: Scalable in silico simulation for human total body exposure prediction using in vitro transdermal and respiratory tract permeability assays
- Modeling of lipogenesis: In Vitro Model Challenges for Skin Lipid Measurements in the Clinic
- 3D bioprinting: Creation of the most sophisticated 3D Bioprinted full skin models, with immunization, pigmentation, vascularization, and oil function to advance cosmetics efficacy testing.
- Systemic Disease modeling for skin: Diabetic Skin: A new target for cosmetic products
- Tissue engineering: Latest advances in tissue engineering: from normal to compromised skin
![]()
 Michael Anthonavage has 26 years of experience in personal care product development and a career spanning background in skin biology, education, and medical technology. Michael has extensive knowledge in product development in personal care product design and specializes in R&D to marketing translation as well as claims validation both in-vitro and in-vivo. He is also an engaging public speaker and product technology advocate with an ability to marry complex ideas and concepts to various consumer needs. Michael is currently the VP of Operations & Technology at Eurofins CRL, Inc. as well as an educator in herbal studies, clinical lab interpretation, product development strategies, physiology, and skin biology. Michael’s previous positions have focused on product development for multi-national corporations in Consumer Products and has held R&D leadership positions at several industry ingredient suppliers where he has championed innovative ingredient portfolios. Michael is currently on the NYC SCC Scientific Advisory Board and has given several lectures for the SCC over the years. He has a variety of publications and patents to his name and continues to be an influential speaker and educator in the personal care, bioinstrumentation, and skin testing arena.
Michael Anthonavage has 26 years of experience in personal care product development and a career spanning background in skin biology, education, and medical technology. Michael has extensive knowledge in product development in personal care product design and specializes in R&D to marketing translation as well as claims validation both in-vitro and in-vivo. He is also an engaging public speaker and product technology advocate with an ability to marry complex ideas and concepts to various consumer needs. Michael is currently the VP of Operations & Technology at Eurofins CRL, Inc. as well as an educator in herbal studies, clinical lab interpretation, product development strategies, physiology, and skin biology. Michael’s previous positions have focused on product development for multi-national corporations in Consumer Products and has held R&D leadership positions at several industry ingredient suppliers where he has championed innovative ingredient portfolios. Michael is currently on the NYC SCC Scientific Advisory Board and has given several lectures for the SCC over the years. He has a variety of publications and patents to his name and continues to be an influential speaker and educator in the personal care, bioinstrumentation, and skin testing arena.
SOFW.com – Interview with Giorgio Dell’Acqua, 2022 NYSCC Chair
Interview with Giorgio Dell’Acqua, 2022 NYSCC Chair
Giorgio Dell’Acqua
2022 NYSCC Chair
Last November the Chapter successfully organized the first ‘back-in-person’ NYSCC Suppliers’ Day after the pandemic break. How was the turnout for attendees and exhibitors?
It was incredible and extremely rewarding to meet face-to face and be reunited with so many members and colleagues. This important event, November 10th and 11th in 2021, attracted 6,807 registrants and 392 exhibitors and they were all eager to find solutions and move forward in their businesses. Noteworthy were the international participants, from 30 countries, who were able to attend despite the travel restrictions.
At the conclusion of this Suppliers’ Day we also had a “Virtual Month of Beauty” that captured even more new registrants and fostered continued engagement.
A big shout to Susanna Fernandes, the 2021 NYSCC Chair, and the entire Suppliers’ Day team for navigating through such unfamiliar territory and being able to bring this edition of Suppliers’ Day to life!
What will the main focus of the 2022 NYSCC Suppliers’ Day on May 3-4, 2022?
The theme this year is “Your Destination for Science & Sustainable Sourcing Solutions.” We are getting back to basics while viewing all chemistry and formulating through a sustainable lens. This will be evident on the exhibit floor and woven throughout the conference program. We will have subject matter experts in the field of digitalization, sustainability, clean beauty and advanced scientific testing, to name a few, presenting during the show and part of our comprehensive educational program. All designed to inspire creativity, ingenuity, and innovation.
NYSCC also formed a Diversity & Inclusion Committee this year that will be involved in programming and activities at Suppliers’ Day to engage attendees in conversation about ideas that provide leadership in the cosmetics community around diversity and inclusion.
We cannot just be focused on the science of the ingredients and formulations without considering the impact of their sourcing both on the environment and society.
What can attendees expect this year?
As the only event in North America for ingredients, formulations, and delivery innovations, NYSCC Suppliers’ Day will have something for everyone.
It will be the best forum in North America for the latest trends, scientific findings, global ingredients, raw materials and solutions that will invigorate formulations and accelerate beauty and personal care product developments.
From end-to-end of the Javits Center, scientific and sustainable sourcing solutions that are impacting product development and brand creations will be discussed, experienced and on full display.
Suppliers’ Day this year will also feature 50+ individual conference sessions and curated educational programs, with 430+ exhibitors with more than 8,000 attendees expected to attend from all over the world. Dynamic areas on the show floor, that once again is expanded into the D Hall at the Javits Center, will provide enhanced experiential learning including the classic Future Chemists Workshop, Presentation Theater, and the return of the INDIE 360 Pavilion. New this year will be a Poster Showcase, featuring the latest scientific findings and ingredients and formulation breakthroughs.
Suppliers’ Day for attendees will help catapult professional advancement and truly be non-stop learning, discovery, and business building.
What will be some highlights of the conference program?
NYSCC continues to grow and enhance the educational component of Suppliers’ Day. This year we are excited to present our hallmark programs with updated content designed to help those involved in formulations, sourcing, and marketing beauty and personal care products achieve their business objectives including:
- Discover Sustainability a series of quick but powerful presentations from leading companies that are successfully implementing clean beauty, green formulation, bio-based and cradle-to-cradle certifications, ethical sourcing, and more.
- Digital Age of Beauty focusing on current strategies and innovations that influence product development, and speed to market. The latest digital tools and techniques that drive, measure and analyze consumer engagement and the demands they set forth will also be presented.
- World of Chemistry delivering a global perspective and discussion on raw materials, solutions, formulation, and regulations. Presenters encompass leading experts from countries and regions that are defining the beauty and personal care landscape.
- Show Floor Presentation Theater complimentary to all attendees, provides insightful, leading-edge supplier presentations and interactive talks.
After last year’s success, INDIE 360 will return and we will be working with IBA (Independent Beauty Association) and other new partners on creating a program that focuses on every angle of the business. A highlight will be brand founders sharing their candid stories and experiences as well as challenges, opportunities and pathways to success. There will also be a spotlight on how INDIE companies are utilizing unique ingredients or innovative ingredient combinations.
PCPC will also be back to present essential content on cosmetic regulation, safety assessment, and quality assurance.
The NYSCC Scientific Advisory Committee will present two conference sessions that take deep dives into topics that are relevant and timely to chemists and R&D teams and the pre-conference SCC CEP Courses will take place on May 2nd.
We also will continue our nurturing and support of the next generation of chemists with the expansion of our Mentor/Mentee Program and the Future Chemists Workshop with even more colleges and universities participating. The co-sponsored SCC & NYSCC Career Development Day will also be part of the event
Of course we will also have our Industry Awards Night celebration after the first day of the show. This year, the finalists of the CEW Beauty Creators Supplier’s Award will be revealed. Industry Awards Night is a great event for renewing partnerships and creating new ones and truly acknowledges the important drivers of innovation and exemplifies Suppliers’ Day core spirit.
Will the event be hybrid?
Yes we will offer a virtual day on May 9th. This will literally be an immersive experience with attendees feeling like they entered the Javits Convention Center in NYC and will give them a 360 degree view of the actual exhibits and expo floor from their desktops or mobile devices.
We also have a platform that makes it easy for exhibitors to provide the same assets and product information for both the live and virtual events and to even schedule appointments with the virtual attendees.
Some of the educational programs offered during the in-person Suppliers’ Day will also be available on May 9th.
Thank you very much for this interview and I hope to see many of your readers at Suppliers’ Day this year. Please visit www.nyscc.org/suppliersday/ for more information and to register.
Sunscreen Monograph Proposed New Rules and its Impact on Formulations – Part III
Sunscreen Monograph Proposed New Rules and its Impact on Formulations-Part III
Introduction
Ever Since the FDA published its proposed rules in February of 2019 reclassifying 14 of the 16 sunscreens approved as Category I to Category II and III, the entire US sunscreen market has been upside down. Most sunscreen manufacturers scrambled to formulate new products incorporating mineral sunscreens like Zinc Oxide and Titanium Dioxide since they were the only sunscreens left as Category I.
Mineral Sunscreens
Although most companies have mineral formulations already, these are mostly targeted to babies and young children. Now the pressure is on to produce mineral sunscreens with high SPF suitable for all consumers and that can compete with organic formulations. This is a task that is not easily achieved, especially with the whitening effect associated with these formulations. Raw material suppliers have been also affected. Polymers, SPF boosters, emulsifiers, and other key ingredients that provided a benefit in organic sunscreen formulations are no longer promoted. Instead, most raw material manufacturers have to promote ingredients that work mainly in mineral formulations.
Market Snapshot for Zinc-Based Formulations
A snapshot of the percent of zinc-based formulations launched globally from 2018 to 2020 (to date) is displayed in Figure I (below). The data shows that the US (34%) leads both Europe (10%) and Asia Pacific (27%) in the percent of new launches of zinc-based sunscreens. This ratio is quite high and shows the effect of the recent scrutiny by the FDA on the safety of sunscreens in the US. In general, the US market is in-line with the European market in terms of customer preference of organic-based versus inorganic-based sunscreen formulations. Historically, inorganic sunscreens were more popular in Asia than the rest of the world due to their impact on skin tone.
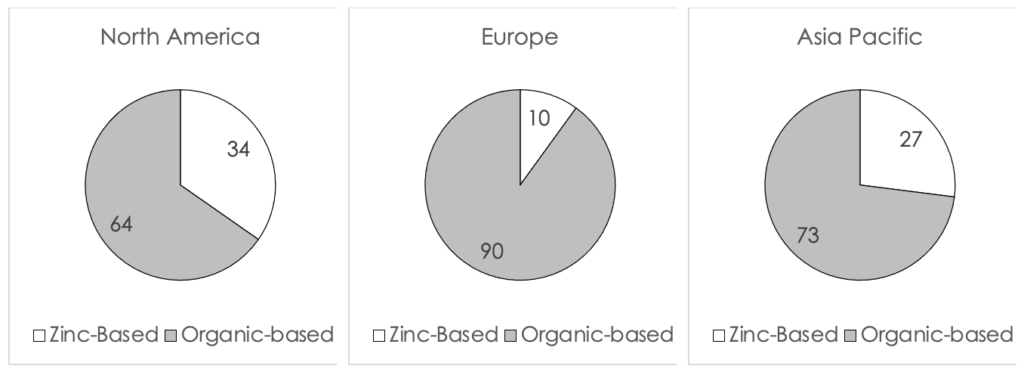
Figure I. A comparison of the percent of global launches of zinc-based formulations from 2018 to 2020 (to date)
The progressions of the percent of new launches from 2018 to 2020 (to date) in the US, Europe and Asia Pacific are displayed in Figure II, III and IV (below). In the US, the number of zinc-based formulations launched in 2020 increased by 7% from 2018. This is a very significant increase considering that many launches in 2020 were postponed due to COVID-19. In Europe the increase was more modest and kept only at 4%, whereas in Asia Pacific the increase was 8%. It can be seen from the data that the global market was affected by the new direction seen in the US market.
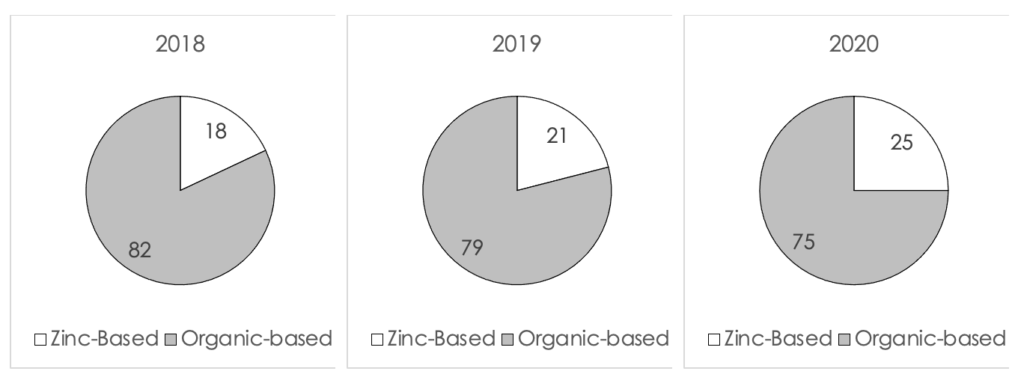
Figure II. A comparison of the percent of launches of zinc-based formulations in North America from 2018 to 2020 (to date)
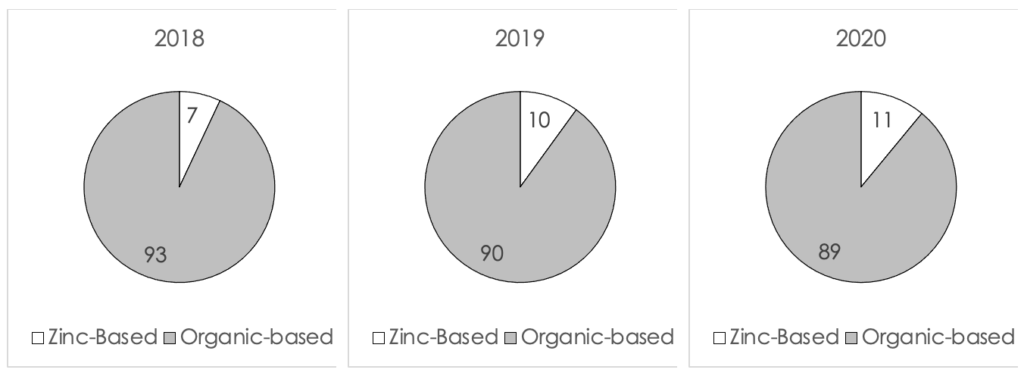
Figure III. A comparison of the percent of launches of zinc-based formulations in Europe from 2018 to 2020 (to date)
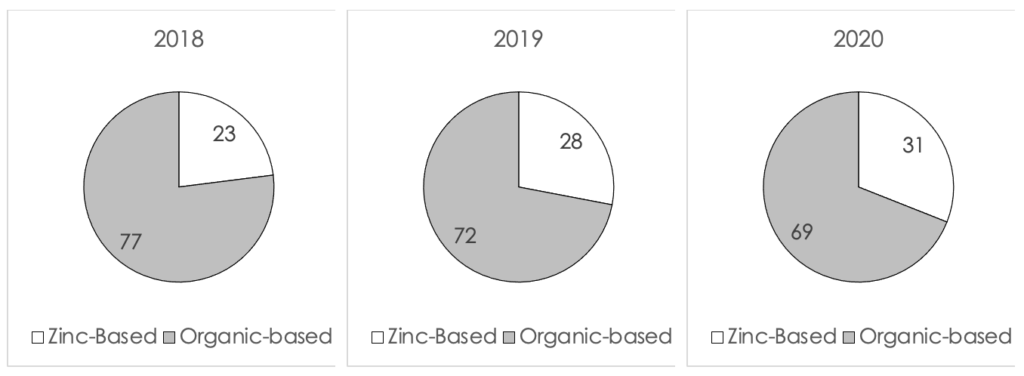
Figure IV A comparison of the percent of launches of zinc-based formulations in Asia Pacific from 2018 to 2020 (to date)
Possible European Actions
To add more complexity to the situation, the European Chemicals Agency, also known as ECHA, is considering classifying most zinc oxide and titanium dioxide sunscreens as microplastics. Since most zinc oxide and titanium-dioxide particles are in the nano range and are typically coated with silicones or other hydrophobic materials, they fit the description of microplastics. This argument is now being debated at the European Union. Considering inorganic sunscreens as microplastics will warrant reformulation of hundreds of formulations worldwide and will have a huge global impact. The impact will be very similar to the one created by the FDA in the US but this one goes in the opposite direction. It will be interesting to see how companies will adapt to all the new regulations. In one aspect, formulators with sunscreen background will be quite in demand and that is a positive outcome for formulation chemists especially during this global crisis of COVID-19. This change will keep all our regulatory experts in demand as well, as someone must decipher what the FDA and ECHA are planning to do in the future. Not to add more complexity, but I am sure China and Australia will come soon with their own guidelines.
Considerations for the Future
For now, the best bet is to be ready and have formulations that will be suitable for the various markets. Global formulations might become a thing from the past.
About the Author

Dr. Hani Fares started his career in personal care studying the effect of solvents on sunscreen chemicals. His interest in skin drug delivery especially from polymeric matrices grew during his graduate work at Rutgers, where he completed his Ph. D. in Pharmaceutics.
Dr. Fares worked at Block Drug and GlaxoSmithKline where he held positions in research and development in the areas of skincare and oral care. After that, he joined L’Oreal where he held several positions of increasing responsibility leading to AVP of skincare. He is currently the Senior Director of skincare and oral care at Ashland Specialty Ingredients. Dr. Fares is the author of many publications, and patents and made many presentations in national and international meetings in the areas of suncare, skincare, and oral care.
A Journey into Color Cosmetics and Lip Product Development
Introduction
The birth of color cosmetics is said to be traced back all the way to Mesopotamia over 5000 years ago where gems and semi-precious gems were ground up and applied to both the lips and the eyelids. In 4000 BCE, Egyptian women would apply Mesdement, a dark grey ore of lead, Galena chemically known as Lead Sulfide, and Malachite (bright blue green paste of Copper minerals) to color their faces for color. They also extracted color from seaweed, Iodine, and beetles to further add to their makeup palette. (1) Somewhere around 3000BCE women in in Greece applied a paste to their face consisting of white chalk, or lead, mixed in with crushed fruits such as Mulberries. They also used lipsticks comprised of clays mixed with red Iron.(1) During the Renaissance Era it was recorded that women use Arsenic in their face powders to replace lead and it wasn’t until the early 1800s that both of these were replaced by what is commonly known today as Zinc Oxide. In 1894 we saw the birth of what is known today as the PCPC, or Personal Care Products Council. Soon thereafter and beginning in the 1900’s the world saw a rapid increase in popularity of color cosmetics as well as the conception of many cosmetic companies commonly known today such as L’Oreal, Max Factor, Avon, and Maybelline to name a few. With this surge came new product forms such as hair dye, mascara, the first liquid nail polish, the powder compact, and in 1928, the very first lip gloss. In the 1940’s leg makeup was developed out of a need of necessity to fulfill the shortage in stockings due to World War II. Over the course of the next 60-70 years the beauty industry would experience a rapid growth spurt in not just technology but also in regulatory bodies that formed the very foundation for what our industry is today.
Color Ingredients
When we speak of color cosmetic formulations, the primary desired end benefit of these products is the same as it was thousands of years ago and that is to impart color to a part of the face to enhance one’s beauty and feel good about oneself. Whether it be a lipstick, foundation, concealer, eye shadow or pressed powder, the color additives are the main constituent and the base formula is what delivers the color and differentiates the product form. These color additives typically consist of pigments, lakes, dyes, and mica and their differences are explained below:
Pigments– Insoluble particles that impart a color to a product. Inorganic pigments are derived from minerals and have good stability to heat and light. Organic pigments, by nature, are carbon based and tend to have a brighter appearance to them. Commonly, pigments are often surface treated and sold in dispersions of various esters and oils to facilitate ease of use in manufacturing and ensure homogeneity of the color.
Lakes– Produced through precipitation of FD&C soluble dyes with metallic salts such as Aluminum salts. Lakes are useful to extend the color range of a shade palette. These ingredients are often surface treated and sold in dispersions of various esters and oils to facilitate ease of use in manufacturing
Dyes-Predominantly water-soluble, these ingredients are used in skin care, body care, fine fragrance, hair care, and color cosmetics. There are some dyes that are oil soluble as well.
Mica– A name given to a group of silicate materials that are used to add shimmer and sparkle
Even though all the ingredients above can be used across the broad category of color cosmetics from eye shadow and pressed powders to lipsticks and foundations, for the purposes of this article we will focus solely on the formulation of lipsticks and liquid lip products.
Physiology of the Lips
The physiology of the lips plays a key role when developing lip products. By nature, the lips are thinner than the skin on the rest of your body and therefore are more sensitive to changes in temperature and texture of a product. Therefore, comfortability and wear are 2 key product parameters that must be properly balanced during development. The lips also do not have hair follicles or oil glands and as a result of this are more prone to drying out especially in the winter months or in low humidity climates. As a result of this, key benefit claims such as hydration and intense moisturization are common for this product type. Finally, the lips do not contain melanocytes found in the skin and as such the color from the lips comes from the blood vessels directly under the surface of the lips.
During aging the lips begin to undergo biological changes like that around other parts of the face and body. The shape of the lips begins to become narrower and longer. They begin to lose volume and color overtime making them appear paler and less vibrant in color and the lines on the lip surface begin to deepen often causing the unwanted side effect of bleeding and feathering. Furthermore, they often present themselves with an unsmooth color tone and appearance.
Environmental exposure can also cause premature aging and thinning of the lips. Chronic UV exposure causes a breakdown in the collagen making the lips appear less full and voluminous. Dehydration from extreme weather conditions or poor health habits can lead to premature and undesirable chafing while exposure to environmental pollutants, i.e., free radicals, and smoking can lead to premature lip lines above the top lip.
Lip Product Formulation
Given the sensitivity of the lips and the desire to keep them young and vibrant looking there are multiple lip products out on the market today each with their own unique benefits and performance. Typically, colored lip products are found in 4 distinct anhydrous forms:
- Lipsticks
- Liquid Lipsticks
- Lip Gloss
- Lip Balms
Traditional lip formulations are composed of emollients, oils, waxes, and colorants with the ingredient ranges varying to deliver a stick or liquid format. Emollients/natural oils such Castor Oil, Lanolin, Shea Butter, are primarily used to help disperse colorants, while providing moisture and smooth application. Structuring agents such as Ozokerite, Polyethylene, and Carnauba and gellants are often used to give the sticks rigidity and stability while film formers are often utilized to improve the transfer resistance and wear properties. Benefit ingredients such as vitamins, natural oils, Hyaluronic acid, fragrance, etc. can all be added as well based on the target audience and desired claims.
In terms of finish, lip products typically are found in multiple styles ranging from matte, satin, pearl and shine with sheer to full coverage levels. From a performance perspective, today’s products are a great evolution to those from a century ago offering intense, continuous hydration, waterproof and transfer proof properties, SPF protection, and even all-day long wear.
Conclusion
Over the years the line between color cosmetics and skin care has become less defined. While the primary function of a color cosmetic is to provide color to the face, lips and eyes, the formulas themselves have become more technologically advanced and multi-faceted. Today’s products offer the consumer benefits beyond color such as anti-aging (wrinkle reduction), sun protection, anti-pollution, reduction of oily and acnegenic skin, minimization of scars, and even tattoo and hyperpigmentation coverage. Additionally, companies continue to innovate in new color cosmetic product forms and test methodologies to deliver aesthetically pleasing, high performance products. For certain, this is a space that will continue to not only innovate but also adapt to today’s complex global challenges while pivoting to consumer needs for years to come!
References
Authors

Peter Konish has been in the industry for 25 years and is currently the Director of the Lip Category, Product Development and Innovation for Coty. Prior to this, he was the Global Director of Technical Operations in the Skin Health division of Johnson and Johnson. In this role Peter was responsible for Process Development, Packaging Development, and External Development/Innovation. Before moving to Technical operations, Peter spent 13 years at NeoStrata in Product Development overseeing the development of numerous Anti-Aging, Prestige Beauty, and Dermatological skin and body care. Peter also spent 9 years at L’Oreal in its Fine Fragrance division working on such brands as Ralph Lauren and Kiehl’s. Peter has a background in polymer chemistry, he has co-authored numerous book chapters and scientific publications, and has been an industry speaker at SCC events and annual AAD meetings.

Jeanine Smith has been in the industry for 19 years with her core focus in color cosmetics. She is currently the Senior Manager of the Lip Category at Coty. Prior to joining Coty, Jeanine was a Senior Manager at Avon Products where she worked in product development for the Eye, Nail and Lip categories.




