The skin barrier function has recently dominated the cosmetic media and consumer market segment. The dermal-epidermal junction (DEJ) is a region within the skin which does not get a lot of face time relative to its high-profile neighbor (epidermis / stratum corneum). The DEJ is the birthplace of the epidermis so it seems reasonable to shed some light on light on this structure, and to ask some questions on why this skincare target may be under promoted for anti-age benefits. For this blog post we will use a marketing style “fact sheet” format to guide us through our chat on “The Dermal-Epidermal Junction”
What it is:
Many are aware of the well-known “brick and mortar” model of the epidermis. That structure (house) needs a foundation for stability and functionality. The DEJ is the foundation of the brick and mortar “house”. The DEJ is composed of four component areas: the basal cell plasma membrane with its specialized attachment devices or hemidesmosomes, an electron-lucent area, the lamina lucida, the basal lamina, and the sub-basal lamina fibrous components, including anchoring fibrils, dermal microfibril bundles, and collagen fibers (1). Hemidesmosomes (HDs) are highly specialized integrin-mediated epithelial attachment structures that make cells firmly adhere to the extracellular matrix by establishing a link between the underlying basement membrane (BM) and the internal mechanical stress-resilient keratin intermediate filament (IF) network (2). The next region progressing downward in the skin is the lamina lucida (LL), it is approximately 30–40 nm in width. This region is directly subjacent to the plasma membranes of basal keratinocytes. The layer underneath the LL is called the lamina densa (LD). This layer of the DEJ is 30-50 nm wide and has biochemical/physical interactions with the extracellular matrix (ECM) of the upper dermis (3).
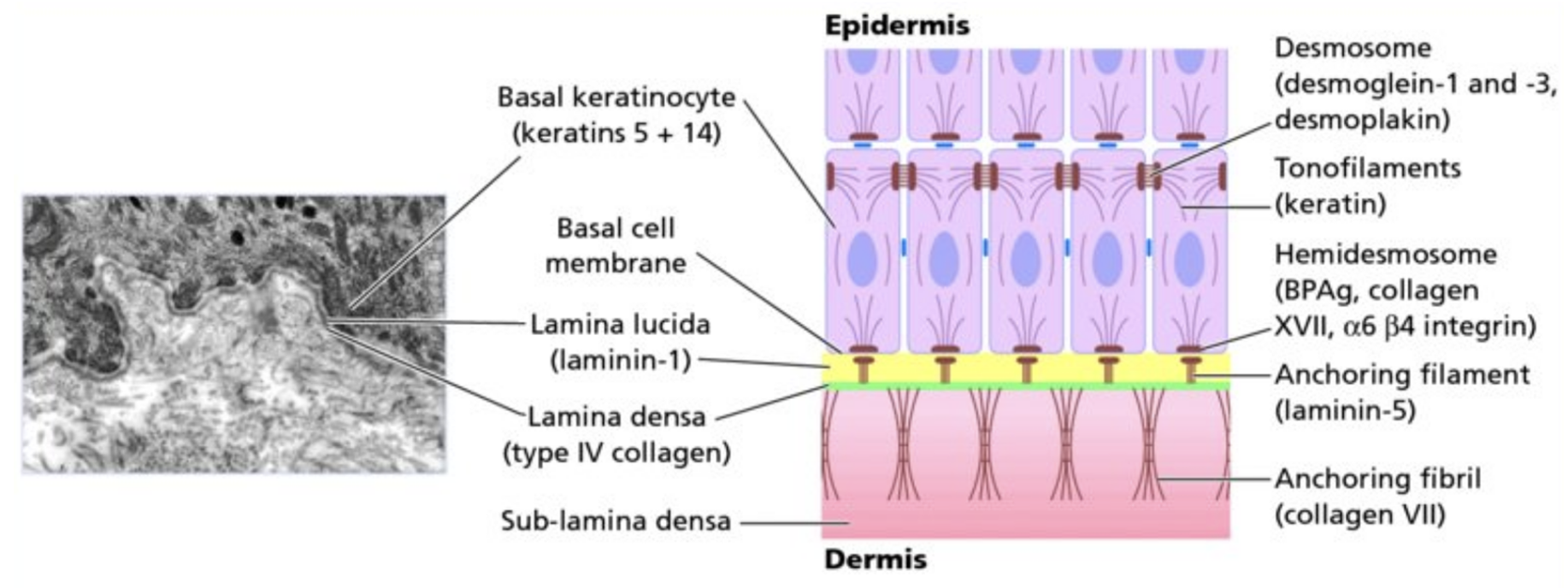 Image Source Here
Image Source Here
What It does:
The dermal-epidermal junction has several functions This area anchors the epidermis to the dermis and is responsible for the exchange of oxygen, nutrients and waste products between the vascularized dermis and the avascular epidermis. This connectivity between the epidermis and the dermis allows for a strong resistance against a possible physical stress (4). The DEJ provides both a structural support to keratinocytes and a specific niche that mediates signals influencing their behavior. The DEJ is also a highly interactive zone acting as a substrate for melanocyte/keratinocyte interactions for melanin distribution as well as a selective permeable barrier for epidermal and dermal cross talk in both directions.
Why isn’t the DEJ a more consistent focus of cosmeceutical product development:
The DEJ forms a fine line between the epidermis and dermis. It is known that the undulating rete ridge area becomes flatter with aging skin. This event lowers the surface area thereby decreasing cellular cross talk and nutrition movement in this region. The dermal capillary structures near the DEJ are a link to the human body and its systemic circulatory network. Could this be a cause of concern for cosmetic products? What if systemic absorption reduces a portion of the active from its sight of action? Another concern may be the potential for a portion of the cosmetic ingredients being moved in the body’s circulatory system. That is a “line in the sand” many companies may not want to cross. The DEJ is complicated, maybe the ability to produce some anti-age benefits in this region is outweighed by the complexity of the task.
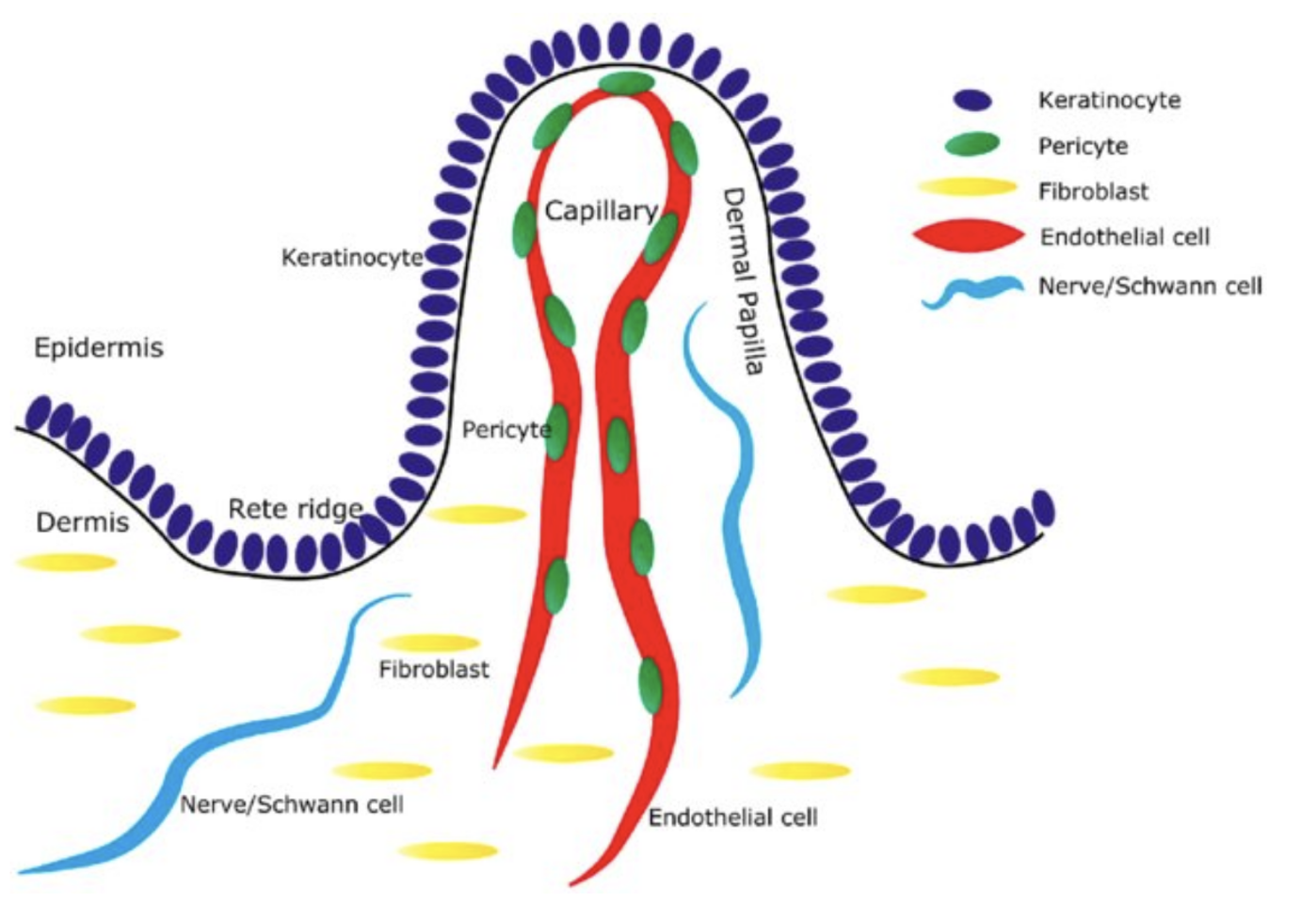 Image Source: Kynan T. Lawlor, Pritinder Kaur: International Journal of Molecular Sciences 16 (12):28098-28107
Image Source: Kynan T. Lawlor, Pritinder Kaur: International Journal of Molecular Sciences 16 (12):28098-28107
How can the DEJ be evaluated or monitored?
If the DEJ is so important, how can we evaluate this area in a noninvasive fashion. One way is to look for a particular protein (Laminin 322) using immunofluorescence (5). Another option to evaluate the DEJ is to use Raman spectroscopy. This technique has been used to evaluate melanin distribution in vivo (6).
What are some DEJ biomarkers of interest for cosmetics?
The dermal-epidermal junction consists of a network of several interacting structural proteins that strengthen adhesion and mediate signaling events (7). Collagen VII stimulates renewal and improves cohesion of the DEJ. Collagen IV is a major constituent in basement membranes. It is involved in maintaining a functional interface between the epidermis and the dermis. Laminin 322 is a key target for DEJ anchoring and cohesion. Peptides have also been identified as opportunities to target to DEJ (8). With this said, there aren’t a lot of cosmetic brands positioning towards the DEJ. The same can be said for raw materials suppliers, I didn’t find a lot of cosmetic materials targeting the DEJ.
In summary, targeting the DEJ can be challenging due to its location in the skin. Caution should be taken as any intended influence of the DEJ from a topical strategy may become systemic due to the proximity of the circulatory/lymphatic vessels. However, that disadvantage may be an opportunity to “feed” the DEJ from a targeted nutritional point of view from within.
References
R A Briggaman, C E Wheeler Jr : The Epidermal-Dermal Junction, J Invest Dermatol, 1975 Jul;65 (1):71-84
Gernot Walko et al. Molecular architecture and function of the hemidesmosome, Cell and Tissue Research 2015; 360(3): 529–544.
Eduardo Calonje , The structure and function of skin : McKee’s Pathology of the Skin, Chapter 1, 1-34.e3
Zhizhong Shen, Rete ridges: Morphogenesis, function, regulation, and reconstruction, Acta Biomaterialia Volume 155, 1 January 2023, Pages 19-34
Lincoln et al. : Gentamicin induces LAMB3 nonsense mutation readthrough and restores functional laminin 332 in junctional epidermolysis bullosa, National Academy of Sciences, PNAS | vol. 115 | no. 28 |
P . Yakimov et al. Melanin distribution from the dermal–epidermal junction to the stratum corneum: non‑invasive in vivo assessment by fluorescence and Raman microspectroscopy, Scientific Reports | (2020) 10:14374
Stephanie Goletz et al. Structural proteins of the dermal-epidermal junction targeted by autoantibodies in pemphigoid diseases, Exp Dermatolactions Dec;26(12):1154-1162. doi: 10.1111/exd.13446.
Sekyoo Jeong et al. Anti-Wrinkle Benefits of Peptides Complex Stimulating Skin Basement Membrane Proteins Expression, Int. J. Mol. Sci. 2020, 21, 73;
![]() About the Author
About the Author
 Marc Cornell, BS. is a consultant at Mar-key Consulting LLC where he services the consumer product industry with innovative product development concepts.
Marc Cornell, BS. is a consultant at Mar-key Consulting LLC where he services the consumer product industry with innovative product development concepts.




 Mythili Nori has worked in the Personal Care industry for over a decade. Her expertise is in Product Claim Substantiation and Data Science. In her current role at BASF, she is responsible for Physical Claim Substantiation & Sensory testing for Hair & Skin Care. Prior to joining BASF, she spent 5 years at TRI/Princeton as a Senior Research Associate, supporting claim substantiation and fundamental research activities for textile and hair surfaces. She earned a Bachelor of Technology in Chemical Engineering from India and received Master of Science in Chemical Engineering at North Carolina Agriculture & Technical State University focusing on purification of drinking water.
Mythili Nori has worked in the Personal Care industry for over a decade. Her expertise is in Product Claim Substantiation and Data Science. In her current role at BASF, she is responsible for Physical Claim Substantiation & Sensory testing for Hair & Skin Care. Prior to joining BASF, she spent 5 years at TRI/Princeton as a Senior Research Associate, supporting claim substantiation and fundamental research activities for textile and hair surfaces. She earned a Bachelor of Technology in Chemical Engineering from India and received Master of Science in Chemical Engineering at North Carolina Agriculture & Technical State University focusing on purification of drinking water.