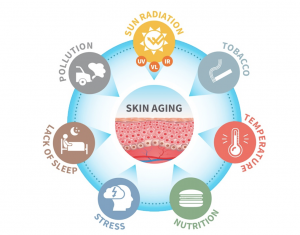
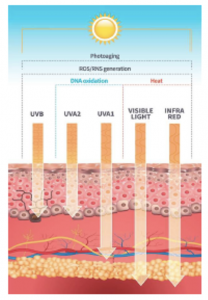
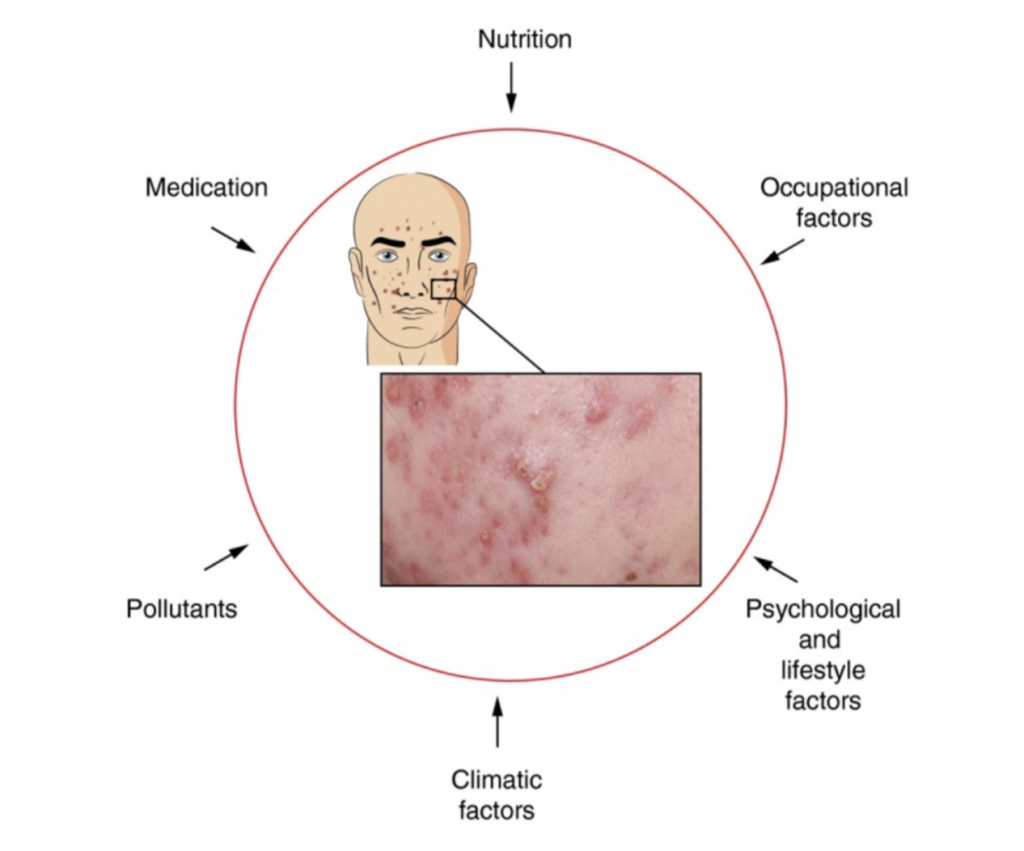
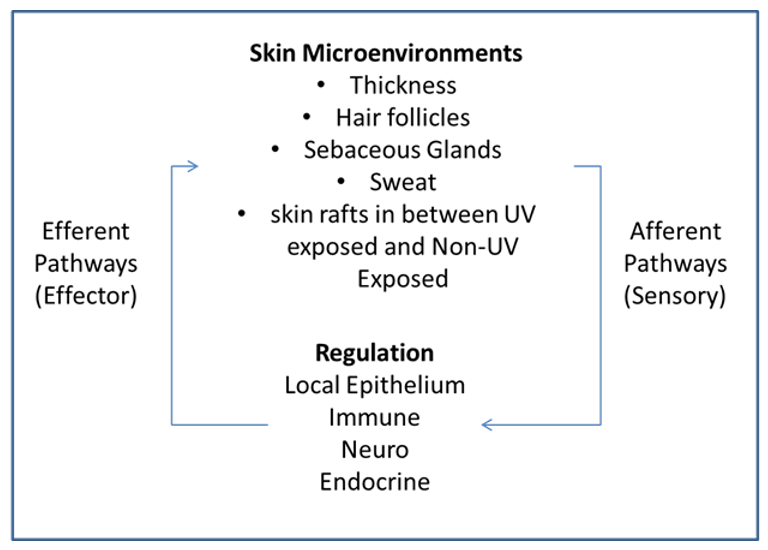
Modern-day formulators relied on polymers to stabilize o/w emulsions much more than surfactants. The introduction of polyacrylic acid-derived polymers many years ago enabled formulators to develop stable emulsions with minimal effort. In realty, formulators used polymers as their primary stabilizers, and they selected the surfactant and esters to tailor the texture and sensorial properties of emulsions. In fact, polyacrylic acid-based polymers enabled steric stabilization of emulsions due to their anionic charge and contributed to the entropic stabilization due to their ability to bind water very efficiently. The art of formulation using the concepts of Hydrophilic Lipophilic Balance (HLB) was almost extinct and was replaced by the fast-paced polymeric stabilization.
In recent years, consumers have been driving the trend of naturality demanding manufacturers to formulate their products with naturally-derived ingredients rather than fossil-based ones. This push towards naturality is forcing formulators to remove their fossil-derived polymers and replace them with naturally-derived counterparts. At the same time, formulators are also replacing their efficient polyoxyethylene-based (POE) surfactants with polyglyceryl based ones, as POE is no longer in vogue with some consumer groups. One can say that formulators who have been spoiled for many years with ease of formulation and guaranteed stability outcome are faced with one of their biggest challenge in recent memory.
The search for an identical, naturally-derived replacement of polyacrylic acid-based polymer has created a frenzy among finished-goods companies and raw material suppliers to try to fill the gap. The first instinct was for formulators to go back and rely on the good old stand-by, xanthan gum. Xanthan is produced by fermentation, so it is considered naturally-derived. It is used in relatively low concentrations and has good yield value. Although xanthan gum has many good attributes, it still has several draw backs. First, its impact on viscosity is minimal and does not build it efficiently. Second, it adds a negative slip and tack to formulations that is quite undesirable. Third, its effect on stability is positive but not quite as good as polyacrylic-based polymers. Formulators need to make several trials before achieving good stability with xanthan gum.
Another stand-by ingredient is starch. Starches have been used to thicken and generate yield in emulsions for many years. An example of a commonly used starch is hydroxypropyl starch phosphate. Starches typically work through a wide pH range (3-9) and have good salt tolerance. However, starches are not efficient thickeners as they have to be used between 1 and 4% w/w in the emulsion to impart stability. When a high level of polymer is used in emulsions it not only reduces the available water for the surfactant to behave properly but it also imparts a certain texture to the formulation which might not be very desirable.
Recently, several companies introduced a variety of gums to stabilize emulsions. Most recently Diutan gum was introduced. Diutan is a high molecular weight polysaccharide (5 MM Dalton) with a relatively low charge density on the backbone. The backbone is made up of four-sugars, namely glucose, glucoronate, glucose and rhamnose and a two-sugar side chain of rhamnose. Diutan seems to be electrolyte tolerant and builds higher viscosities than xanthan gum when combined with a low level of electrolytes. However, it does not build enough viscosity on its own based on literature.
Several manufacturers tried combining several natural gums to achieve good emulsion esthetics and stability. One manufacturer combined xanthan gum, with sclerotium gum, and pullulan. Other manufacturers are combining acacia and gellan gum, xanthan and guar gum, as well as acacia and xanthan gum. Such combinations could be good options, but finished-goods formulators tend to lean more towards single ingredient substitutes as they do not crowd the ingredient label and offer greater flexibility in formulation. In addition, many of these combinations have similar esthetics and do not offer a robust stability profile.
More recently, a new grade of cellulose gum was launched. This type of cellulose can suspend and has a yield value which separates it from common cellulose gums. This readily biodegradable polymer was used in stabilizing O/W emulsions made with organic sunscreens as well as inorganic sunscreens. In one example, formulators were able to develop an O/W inorganic sunscreen formulation containing 20% w/w zinc oxide. The polymer showed great synergies with currently available, naturally derived polymers like xanthan gum and hydrophobically modified hydroxyethylcellulose. In addition, the polymer appeared to yield viscosities similar to the one achieved by polyacrylic acid when used alone or in combination with other naturally-derived thickeners.
As a formulator, I am still hopeful in finding an exact replica of a polyacrylic acid type polymer that is naturally derived, biodegradable, efficient, low cost and with good esthetics. At one point reality will sink in, and will realize that such polymer will not exist. The mere fact is that the chemical make-up of the backbone of the polymer will be different, and unlike polyacrylic-acid based polymers, the natural ones will not be crosslinked. Instead, many of the naturally-derived ones are linear polymers with some branching. In this fast-paced environment, formulators will have to adapt and sharpen their formulation skills. They will use their creativity and I am sure will create amazing textures with the toolbox they currently have until new technology is introduced or new market trends appear.
Dr. Fares started his career in personal care studying the effect of solvents on sunscreen chemicals. His interest in skin drug delivery especially from polymeric matrices grew during his graduate work at Rutgers, where he received his Ph. D.
![]()

Dr. Fares started his career in personal care studying the effect of solvents on sunscreen chemicals. His interest in skin drug delivery especially from polymeric matrices grew during his graduate work at Rutgers, where he received his Ph. D.
Dr. Fares worked at Block Drug and GlaxoSmithKline where he held positions in research and development in the areas of skincare and oral care. After that, he joined L’Oréal where he held several positions of increasing responsibility leading to AVP of skincare. He is currently the Senior Director of skincare and oral care at Ashland Specialty Ingredients. Dr. Fares is the author of many publications, and patents and made many presentations in national and international meetings in the areas of suncare, skincare, and oral care. Dr Fares chairs the NYSCC scientific committee and has won multiple awards in the areas of sun care and polymer chemistry.

 Dr. Amber Evans is a cosmetic industry professional with over a decade of experience in research and innovation. In her current position as Senior Manager of Product Development at Moroccanoil, she leverages her technical expertise to help drive the global launch of prestige hair & body care products. Prior to Moroccanoil, she worked as a development scientist at ingredient supplier BASF Corporation, where her contributions spanned multiple market segments, including hair, body, and oral care. She also previously supported initiatives such as upstream research for hair colorants and clinical testing for skin/shave care applications at Procter & Gamble.
Dr. Amber Evans is a cosmetic industry professional with over a decade of experience in research and innovation. In her current position as Senior Manager of Product Development at Moroccanoil, she leverages her technical expertise to help drive the global launch of prestige hair & body care products. Prior to Moroccanoil, she worked as a development scientist at ingredient supplier BASF Corporation, where her contributions spanned multiple market segments, including hair, body, and oral care. She also previously supported initiatives such as upstream research for hair colorants and clinical testing for skin/shave care applications at Procter & Gamble.